Nitrate In The Reef Aquarium

By Randy Holmes-Farley
Introduction
Nitrate is an ion that has long dogged aquarists. It is typically formed in aquaria through the digestion of foods, and in many aquaria it builds up and can be difficult to keep at natural levels. In the past, many aquarists performed water changes with nitrate reduction as one of the primary goals. Fortunately, we now have a wide array of ways to keep nitrate in check, and modern aquaria suffer far less from elevated nitrate than they have in the past. In some case, aquarists have even found that they have reduced it too far, and nitrate dosing can even be beneficial.
Elevated nitrate is often associated with algae, and indeed the growth of algae is often spurred by excess nutrients, including nitrate. The same can be said for other potential pests in aquaria, such as dinoflagellates. Nitrate itself is not acutely toxic at the levels usually attained in aquaria, at least as it is so far known in the scientific literature, but elevated levels do seem detrimental to the health of fish. Additionally, elevated nitrate can spur the growth of zooxanthellae in corals, which can darken corals and may decrease the growth rate of a host coral.
For these reasons, most reef aquarists strive to keep nitrate levels down. Some are very successful, and others are not. This article provides some background on nitrate in the ocean and in aquaria, and describes a number of techniques that aquarists have successfully used to keep nitrate levels down to more natural levels in reef aquaria.
What is Nitrate?
Nitrate is a negatively charged ion, NO3–. It consists of a central nitrogen atom and three oxygen atoms attached to it. The negative charge is spread out equally over the three oxygen atoms, making them all identical. It is generally very soluble in seawater.
Nitrate in the Ocean
Nitrogen takes many forms in the ocean, one of which is nitrate.1 Other forms include dinitrogen (N2), ammonia (NH3/NH4+), nitrite (NO2–), and a myriad of nitrogen-containing organic compounds. Of the inorganic species, nitrate is often, but not always, the highest in concentration. Concentrations in the ocean vary considerably from location to location, and also with depth.2 Surface waters are much lower in concentration due to scavenging by various organisms, and are often less than 0.1 ppm nitrate (note that all concentrations in this article are in ppm nitrate ion, and not in ppm nitrate nitrogen). Deeper ocean waters typically range from 0.5 to 2.5 ppm nitrate. Surface regions where upwelling of deeper water takes place will also have these higher values.
Most of the nitrate present in the ocean results from the recycling of organic materials. The degradation of plankton, for example, provides nitrate. This can be shown in a simplified chemical equation describing what happens when organic “food” is digested with oxygen:
(CH2O)106(NH3)16(H3PO4) + 138 O2 → 106 CO2 + 122 H2O + 19 H+ + PO43- + 16 NO3–
which in words reads as:
plankton + oxygen → carbon dioxide + water + hydrogen ion + phosphate + nitrate
Note that this process consumes alkalinity (the H+ being produced shows this). So when nitrate is accumulating in a reef tank, alkalinity is being depleted. Production of 10 ppm of nitrate will deplete about 0.16 meq/L (0.45 dKH) of alkalinity. If this nitrate is removed by water change, that alkalinity is lost forever. If the nitrate is taken up by an organism (algae, coral, bacteria, etc.) and used, then all of that alkalinity is returned to the system (see equations below showing this fact).
Other sources of nitrogen to the ocean are volcanic emissions (mostly as ammonia), fixing of N2 by blue-green algae (also known as cyanobacteria), and run-off from land. All of these become part of the nitrogen cycle, and a portion will end up as nitrate.
Marine Organisms That Use Nitrate
A wide variety of organisms are capable of absorbing nitrate with which they synthesize a host of nitrogen-containing organic molecules, such as proteins and DNA.1 Nitrate is primarily used by microorganisms (such as bacteria) and those organisms that get much or all of their energy from photosynthesis, including algae, corals and sea anemones.
In some circumstances and for some organisms that use nitrate, elevated levels of nitrate can result in increased growth. Microalgae in reef aquaria can grow faster in higher nitrate if they have adequate phosphorus, iron, light, etc. Large organisms are impacted as well. Shoalgrass (Halodule wrightii) and widgeongrass (Ruppia maritime) grow faster in elevated nitrate (0.6 ppm nitrate) than in typical ambient seawater nitrate levels (<0.1 ppm nitrate).3 The various sea grasses have systems for active uptake of nitrate from both leaves and roots, but still cannot get all they “want” from normal seawater.4
Marine bacteria5, phytoplankton6,7, and macroalgae7,8 have also been shown to increase growth rates with elevated nitrate. In other cases, elevated nitrate does not increase growth. In these cases, other factors are limiting, such as phosphate, iron, or light. The growth of the sea grass Zostera marina, for example, is not enhanced by increased nitrate, with growth more often being limited by light.3,4,9
Obviously, some of the organisms that grow faster in water with elevated nitrate are not necessarily those that aquarists most prefer. Beyond the obvious concerns about microalgae, dinoflagellates seem to increase growth as the nitrate and other nutrients increase, up to at least 16 ppm.10 It may, however, come as a comfort to some aquarists to know that the pest anemone Aiptasia pulchella can only take up nitrate under starvation conditions, and even then not very well.11,12
Fish, it seems, are not very sensitive to nitrate. Most researchers find little toxicity13. One group that studied a variety of species of fish larvae report:
“Judging from its effect on 1st-feeding, unionized NH3 is a potential hazard in the rearing tank; NO2– and NO3– are nontoxic at levels likely encountered in practical marine fish culture.”14
Still, many hobbyists report that their fish appear less healthy when they have allowed nitrate levels to get excessively high (over 50 ppm). Whether that is actually due to nitrate or something else about the water that is coincident with the nitrate rise is unknown.
Finally, the addition of excess nutrients to natural coral reefs has been blamed for a general transition from corals to turf and macroalgae,15 but what role nitrate plays relative to other nutrients (such as phosphate) is not always clear.
Effects Of Elevated Nitrate In Aquaria
In addition to the concerns described above relating to the growth of potentially undesirable organisms that may be promoted by elevated nitrate (especially algae and dinoflagellates), corals can be impacted by nitrate. Many corals may not be bothered by elevated nitrate, or may even grow more rapidly with the readily available nitrogen. But in certain corals, especially those that calcify, there may be negative effects from elevated nitrate.
In most cases where nitrate levels have been examined in relation to the growth of calcareous corals, the effects have been reasonably small, but significant. Elevated nitrate has been shown to reduce the growth of Porites compressa (at less than 0.3-0.6 ppm nitrate),16,17 but the effect is eliminated if the alkalinity is elevated as well (to 4.5 meq/L). One explanation is that the elevated nitrate drives the growth of the zooxanthellae to such an extent that it actually competes with the host for inorganic carbon (which is used both in photosynthesis and in skeletal deposition). When the alkalinity is elevated, this competition no longer deprives the host of needed carbon.17
A second study on Porites porites and Montastrea annularis tends to support this hypothesis. They showed that elevated nitrate caused an increase in photosynthesis, in the density of zooxanthellae, and in their chlorophyll a and c2, and total protein, while skeletal growth decreased considerably.18 This effect may not always be true, however, since elevated nitrate does not appear to have decreased calcification in Acropora cervicornis (though the experiments were carried out under very different conditions).19
A third study, on Acropora pulchra, showed the increased nitrate increased zooxanthellae levels and the calcification rate.20 One other study 21, on Porites cylindrical, has reported that elevated nitrate (0.9 ppm) did not increase the rate of photosynthesis or zooxanthellae density, but actually decreased it, contrary to the other studies. They do not provide an explanation of why their results were different, though they indicated that the corals may have been expelling zooxanthellae, which would confound some of the results. Additionally, all of the corals in the study were stressed in that they lost significant biomass during the study compared to when first collected in the wild. Because of that effect, I do not put much faith in how this study may relate to aquaria where corals are growing rapidly.
Overall, it seems that elevated nitrate may spur zooxanthellae levels in corals, and aquarists may notice this as a darkening (browning) of the corals since zooxanthellae are golden brown. Whether there is a growth rate effect, and in what direction, may depend on the coral and the other conditions in the aquarium.
Measuring Nitrate In Aquaria
Nitrate is fairly easily measured in marine aquaria at levels higher than about 0.5 ppm using test kits. Aquarists sometimes get different results from different test kits, however, even when testing the same water sample. For this reason, I suggest that aquarists look to the effects on their aquarium at least as much as a test kit result when evaluating whether nitrate reduction methods are adequate. Sticking with one brand of kit may also be important. Even if it is relatively inaccurate, monitoring the tank with one kit will at least allow trends to be monitored, while switching between multiple kits might leave the aquarist not knowing whether nitrate is rising, falling, or remaining steady.
Sources Of Nitrate In Reef Tanks
The primary source of nitrate in reef aquaria is food added to the system. All proteins contain nitrogen, as do a wide variety of other biomolecules. When metabolized, much of this nitrogen can end up as nitrate. Fish and other organisms excrete ammonia after digesting food, for example, and nitrifying bacteria quickly convert ammonia into nitrite and then into nitrate. Other inputs can include the die off of organisms, which degrade in a fashion similar to that shown above for plankton.
Finally, the use of unpurified water can lead to significant addition of nitrate to aquaria. In the United States, drinking water is permitted to contain up to 44 ppm nitrate. Daily addition of such water to replace evaporated water will provide a significant amount of nitrate. In many municipal water systems, however, the level of nitrate is much lower. In my water supply, the level is typically only 0.1 ppm nitrate.
Lowering Nitrate In Aquaria
The bottom line for many aquarists is that they have nitrate levels in their aquaria that are higher than they prefer. I strive to keep the nitrate levels in my aquaria below 1 ppm and preferably, less than about 0.5 ppm. That said, I rarely test for nitrate, and gauge nutrient reduction on cues from the aquarium, such as the growth rate of algae on the glass.
This section outlines a variety of actions that can be taken to reduce nitrate levels in aquaria. Note that I don’t include any discussion of water changes, though obviously they work to some extent. I have discussed that effect extensively in a previous article. The problem with water changes is that it is very hard to reduce the nitrate concentration to natural levels in that fashion alone unless the system is constantly flushed with clean water. Nevertheless, it does do a good job of limiting the accumulation of nitrate.
The first action in reducing nitrate is to measure nitrate with a quality test kit to understand the magnitude of the problem and to be able to track progress. Then follow one or more of the actions below and monitor the nitrate over time to see if it is helping.
1. Inputs Of Nitrogen To The Aquaria
If you are overfeeding, stop. I’m not, however, suggesting that folks starve any organisms in their aquaria for the sake of reducing nitrate levels. Bear in mind that it really doesn’t matter from the perspective of the ultimate nitrate delivery to the water column whether the food is eaten by a fish or not. Nearly all of the nitrogen in the food will end up in the water column, whether digested by a fish, a coral, or bacteria and other microorganisms. Fortunately, there are better options available for nitrate control than starving the tank.
If you are using tap water, test it for nitrate to see if it is a source, and if so, purify it first. A reverse osmosis/deionizing system( RO/DI) is best for a variety of reasons, but a simple system employing RO alone or DI alone system will likely be adequate for this purpose.
2. Nitrogen Export by Skimming
Skimming alone does not usually permit aquarists to eliminate a nitrate problem, but it can be a significant help, and also has other benefits, such as aeration and removal of organics and phosphorus. The reason skimming works for nitrate is not because nitrate is skimmed out (it is not), but rather that organic molecules in the water that contain nitrogen can be removed before they break down, releasing inorganic nitrogen. Many proteins, for example, are readily skimmed out, and they all contain a large amount of nitrogen. But this mechanism also shows why skimming alone is often inadequate: it can do nothing for ammonia excreted by organisms that is converted into nitrate by nitrifying bacteria.
3. Nitrogen Export By Growing And Harvesting Macroalgae or Turf Algae (or Any Other Organism of Your Choice)
Growing and harvesting macroalgae or using an algal turf scrubber (ATS) can be a very effective way to reduce nitrate levels (along with other nutrients) in reef aquaria. In my reef system, where I have large, lit refugia to grow the macroalgae Caulerpa racemosa, these algae are clearly a large nutrient export mechanism. Aquaria with large amounts of thriving macroalgae or turf algae may avoid microalgae problems. Whether the reduction in nitrate is the cause of the microalgae reduction is not obvious; other nutrients can also become limiting. But to reef aquarists with a severe microalgae problem, the exact mechanism may make no difference. If rapidly growing macroalgae absorb enough nutrients to keep the nitrate concentrations in the water column acceptably low, and at the same time keep microalgae under control, most reefkeepers will be satisfied.
For those interested in knowing how much nitrate is being exported by macroalgae, this free PDF article in the journal Marine Biology has some useful information. It gives the phosphorus and nitrogen content for nine different species of macroalgae, including many that reefkeepers typically maintain. For example, Caulerpa racemosa collected off Hawaii contains about 5.6% nitrogen and 0.08 % phosphorus by dry weight. Harvesting 10 grams (dry weight) of this macroalgae from an aquarium would be the equivalent of reducing the nitrate content by 2.5 grams, or 10 ppm in a 67-gallon aquarium. All of the other species tested gave similar results (plus or minus a factor of two). Interestingly, using the same paper’s phosphate data, this export would also be the equivalent of removing 24 mg of phosphate from the water column. That amount would reduce the phosphate concentration from 0.2 ppm to 0.1 ppm in a 67-gallon aquarium.
4. Deep Sand Beds
Deep sand beds can develop low oxygen regions where nitrate is used by bacteria. They use it to metabolize organics that randomly diffuse into the sand from the water column. When the oxygen gets depleted in the sand, the bacteria can still oxidize the organics available by using nitrate instead of O2. In this situation, nitrate acts as an electron acceptor (e.g., an oxygen source) in place of oxygen (O2). The end result is that nitrate is converted into N2, and the N2 blows off of the tank to the atmosphere. The reactions that take place can be complex.22 In oxygen-containing environments, the reaction looks very similar to that shown above for plankton (ignoring phosphorus here):
organic + 175 O2 → 122 CO2 + 16 NO3– + 16 H+ + 138 H2O
where organic stands for a typical organic material ((CH2O)80(CH2)42(NH3)16) that is being metabolized. In the absence of O2, and taking the nitrogen species completely to N2 (which may happen in several reaction steps), we have the following overall reaction:
organic + 124 NO3– + 124 H+ → 122 CO2 + 70 N2 + 208 H2O
It can be seen that the process above produces alkalinity (by consuming H+). In fact, it is the exact same amount of alkalinity that was depleted when the nitrate was originally formed from foods, so the net effect of the nitrogen cycle on alkalinity is zeroed out.
In many aquaria, the process takes place to an extent sufficient by itself to keep nitrate at levels below 0.5 ppm. In others, it has not been adequate. Success may depend on the size of the bed, its composition (sand type, particle size distribution, depth, and life forms in it), and the demands put on it in terms of nitrate processing. It is rarely discussed by aquarists, but organics are critical for this process as well, and some aquaria may have more or less organic matter in the water (due to use of things like skimming or granular activated carbon) and this, in turn, can impact the nitrate conversion capability of a sand bed.
This process also happens in the pores of live rock, and in a variety of other environments in a reef aquarium.
5. Filters Designed To Facilitate The Nitrogen Cycle.
Filters such as trickle filters using traditional bioballs do a fine job of processing ammonia to nitrite to nitrate, but do nothing with the nitrate. It is often non-intuitive to many aquarists, but removing such a filter altogether may actually help reduce nitrate. Consequently, slowly removing them and allowing more of the nitrogen processing to take place on and in the live rock and sand can be beneficial.
It is not that any less nitrate is produced when such a filter is removed, it is a question of what happens to the nitrate after it is produced. When nitrate is produced on the surface of impermeable media such as bioballs, it mixes into the entire water column, and then has to find its way, by diffusion, to the places where it may be reduced (inside of live rock and sand, for instance).
If it is produced on the surface of live rock or sand, then the local concentration of nitrate is higher there than in the first case above, and it is more likely to diffuse into the rock and sand to be reduced to N2.
In a reef aquarium with adequate live rock, there is little use for a trickle filter, so in general they can be safely removed.
6. Carbon Denitrators
There are a variety of different commercial systems available, none of which are especially popular in the United States at this time. However, they can do a good job of removing nitrate and some aquarists like them.
In one of these types of systems, a carbon source is added to a portion of tank water in a low oxygen environment. In many cases, the carbon source is methanol. The methanol is mixed with aquarium water in a controlled situation (such as fluid pumped through a coil) and the methanol is consumed by bacteria that use nitrate as an electron acceptor instead of oxygen:
12 NO3– + 10 CH3OH + 12 H+ → 10 CO2 + 6 N2 + 26 H2O
The end result is that nitrate is removed from the aquarium. The typical drawback to such a system is the need for careful control over the conditions, and the consequent complexity that often accompanies such a reactor. Note again that this process returns the alkalinity (by consuming H+) that was lost in the production of the nitrate originally.
This method is similar to organic carbon dosing, but is localized inside of a reactor. Such localization can have advantages (less chance for cyanobacteria to be driven to grow more by consuming the organic), but it is a bit harder to accomplish technically than simple dosing of organics to the aquarium (described below).
7. Biopellets
Biopellets are typically organic polymers (often PHB, polyhydroxybutyrate; shown below)):
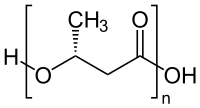
Polyhydroxybutyrate
These polymers can very slowly degrade in sterile water, but in an aquarium, bacteria settle on them and release enzymes that chop up the polymers into smaller bits (such as hydroxybutyric acid, the building block of this polymer) much faster than the simple hydrolysis reaction would proceed on its own. PHB is a natural energy storage molecule for many bacteria, and so some bacteria are accustomed to making it and degrading it internally. Some bacteria earn a living by being ready to degrade such polymers when they encounter them outside their bodies (for example, from other bacteria that have died) and these bacteria release enzymes into the water to break down the polymer pellet. They then take up the released hydroxybutyric acid and metabolize it to gain energy.
In an aquarium setting, bacteria will coat the pellets, and digest much of the polymer themselves, using up nitrate and phosphate in the process as they grow and expand their tissues. Some of the released hydroxybutyric acid will make its way into the bulk water, so bacteria can also grow remotely from the pellets (including cyanobacteria). Consequently, it is not as confined of a process as is a carbon denitrator, even though it is usually carried out in a flow reactor.
This method can use slightly more nitrate than a proportional amount of phosphate if the bacteria form a thick enough layer on the pellet. In that case, the bacteria on the bottom (the pellet surface) can be oxygen limited, and may use nitrate as an electron acceptor (source of oxygen) as happens in deep sand beds, in addition to the N and P used to make their body tissues.
8. Sulfur Denitrators
In these systems, bacteria use elemental sulfur and produce N2 from the sulfur and nitrate according the following equation (or something similar):
2 H2O + 5 S + 6 NO3– → 3 N2 + 5 SO42- + 4 H+
The production of acid (H+) in this reactor can tend to reduce the aquarium alkalinity. It has also been suggested to pass the effluent of such a reactor through a bed of aragonite to use the acid (H+) produced to dissolve the calcium carbonate, and thereby provide calcium and alkalinity to the aquarium. While that is a fine idea, it doesn’t add much calcium and alkalinity to most aquaria.
To estimate the magnitude of the effect, we start with a liberal estimate of how much nitrate might be removed. Say 10 ppm of nitrate per week.
10 ppm nitrate = 0.16 mmole/L of nitrate
Since 4 moles of H+ are produced for every 6 moles of nitrate consumed, this will produce
0.107 mmoles/L of H+ per week
How much calcium this could produce?
Assume that it takes one proton to dissolve one calcium carbonate:
CaCO3 + H+ → Ca2+ + HCO3–
Clearly, this is a substantial overestimate because much of the acid will be used up driving the pH down to the point where CaCO3 can even begin to dissolve. Consequently, we have an upside limit of 0.107 mmoles of Ca2+ per week. Since calcium weighs 40 mg/mmol, that’s 4.3 ppm Ca2+ per week.
For comparison, an aquarist adding 2% of the tank volume in saturated limewater daily is adding on the order of 16 ppm of calcium per day. Consequently, this method may not be especially useful for maintaining calcium. Additionally, the acid produced will have a long term lowering effect on the alkalinity. In fact, it is double dipping on the alkalinity depletion since alkalinity is consumed when the nitrate is produced, and again when it is removed in the denitrator. So if you use a sulfur denitrator, be sure to monitor the alkalinity in the aquarium.
9. Organic Carbon Dosing
Organic carbon dosing involves adding a soluble organic compound to the aquarium which spurs bacterial growth. Typical organics used can be ethanol (as vodka), acetic acid (as vinegar), calcium acetate (as lime saturated vinegar), sugar (sucrose) and many others. Vodka and vinegar are by far the most popular. I use vinegar.
These organic molecules can be used by many organisms, including corals, but the main intent is to drive bacterial growth. To grow, the bacteria need a source of nitrogen and a source of phosphate, and a large portion of these they remove directly from the water. The bacteria may grow out of sight (inside live rock or sand, in refugia, in tubing, etc.). They may also grow in globs in the display tank. They have to grow somewhere. If they become unsightly, try dosing a different organic that may drive a different set of species that may grow in a different location. I’ve had them often seem to grow on GAC (granular activated carbon media) in a canister filter I previously used, allowing relatively easy export by rinsing the GAC once every couple of weeks.
I’ve never heard any plausible argument why dosing multiple organics at once is desirable, but many people do it and there is likely no harm in doing so. The idea that multiple organics drive a diversity of bacterial species is just speculation, and even if true, I don’t see the benefit.
The bacteria themselves can then be skimmed out, or used as a food for filter feeders, or both (most people probably have both to some extent, unless they do not use a skimmer). The bacteria may grow partly in low O2 regions (such as in sand or rock) and partly in highly oxygenated environments. Since metabolism in low O2 regions uses relatively more nitrate than phosphate compared to metabolism in a high O2 environment, the relative amounts of nitrate and phosphate reduction an aquarists observes may vary from system to system.
Nitrate is always reduced to a greater extent than phosphate simply because bacteria need a lot more nitrogen than phosphorus, but metabolism of organics in low O2 regions may skew it even more, and sometimes can leave the aquarium with little nitrate and an excess of phosphate that they bacteria don’t “want”. In such a case, a phosphate binder might usefully export this remaining phosphate. Alternatively, some aquarists have dosed nitrate directly to the aquarium to allow the residual phosphate to be consumed.
These linked articles describe vinegar and vodka dosing in more detail.
One potential drawback that may have played a role in some tank problems is that the bacteria that thrive when organic molecules are dosed may be benign (and appear to be in almost all cases), but might actually be pathogenic in others. That is, the added organics may enhance bacterial infections if those bacteria causing the infection (of fish, corals, etc.) are able to take up the added organics and use them to grow faster. I think this risk is low, but it may be real. If you have unexplained problems that might fit this description, and are organic carbon dosing, try not dosing for an extended period.
A second potential drawback of organic carbon dosing is the potential for proliferation of unsightly cyanobacteria in the display tank. There are many species of cyanobacteria, and some can consume the organics we add in this method. If they become a primary consumer, then something may need to be done, such as switching to a different organic compound to dose, or reducing phosphate with a binder such as GFO (granular ferric oxide).
10. Nitrate Absorbing Solids
Various aluminum oxide and zeolite products have been sold to aquarists for many years for the purpose of binding nitrate out of the solution. Kent’s nitrate sponge is one example. I’ve not tested any myself. Many aquarists report that it does work, but takes a long time and a lot of material.
11. Denitrification Media
There are a variety of porous ceramic media which have internal pores that act much in the way live rock does. Some of these are fairly new to the market (e.g., Cermedia MarinePure blocks) and some have been around a while (e.g., Seachem Matrix). They provide an area where denitrification can take place due to it potentially becoming low in oxygen, and hence like the inside of a deep sand bed or live rock. Some users report great success with these and they are apparently very effective in maintaining low nitrate in many aquaria. One possible drawback that is yet to be really understood is the potential for some of these to release aluminum to the water, which may have toxic effects. How significant this is remains to be established. It may be no concern at all.
12. Polymers And Carbon That Bind Organics
These binders are similar to skimming in that they remove organics from the system, preventing them from degrading and contributing to the organic load. Examples are Purigen by Seachem and Poly-Filters by Poly-Bio-Marine. I’ve not used any of these for this purpose, and have not heard of others significantly reducing elevated nitrate levels with them, but they would accomplish this goal and reducing organics is generally desirable anyway.
Summary
In the past, elevated nitrate was something that many aquarists accepted as a fact of life in keeping marine aquaria. Now, with many ways of reducing nitrate readily available, most aquarists can (and probably should) strive to keep nitrate to more natural levels. With the new methods available, a growing number of aquarists have actually had problems from too little nitrate, so maintaining an appropriate level can be a bit of a balancing act.
I have chosen to keep nutrients low by:
1. Growing and harvesting macroalgae (currently, Caulerpa racemosa) from several large refugia.
2. The refugia are mostly rock-filled to allow significant surfaces for denitrification.
3. I dose vinegar (~175 ml per day to a 120 display with ~300 gallons total system volume) during the daylight hours just upstream of the first refugium.
4. I skim and use GAC (granular activated carbon) for organic removal before it can break down.
5. I also use GFO, but I expect it has almost no impact on nitrate levels.
One thing I do not do is remove particulates (except by skimming) and so detritus collects on the bottoms of my refugia and sumps in a thick layer Those may be a big source of nutrients over time, but the export methods I use apparently can handle the load.
Regardless of what methods you prefer, nitrogen export ought to be one of the design considerations in any reefkeeping setup.
Happy Reefing!
For more Reef Chemistry questions or topics please visit our Reef Chemistry forum here.
References
- The Complete Nitrogen Cycle by Randy Holmes-Farley Aquarium Frontiers http://www.fishchannel.com/saltwater-aquariums/aquarium-frontiers/nitrogen-cycle.aspx
- Chemical Oceanography, Second Edition. Millero, Frank J.; Editor. USA. (1996), 496 pp. Publisher: (CRC, Boca Raton, Fla.)
- Comparative effects of water-column nitrate enrichment on eelgrass Zostera marina, shoalgrass Halodule wrightii, and widgeongrass Ruppia maritima. Burkholder, JoAnn M.; Glasgow, Howard B., Jr.; Cooke, Jacob E. Dep. Bot., North Carolina State Univ., Raleigh, NC, USA. Marine Ecology: Progress Series (1994), 105(1-2), 121-38.
- Review of nitrogen and phosphorus metabolism in seagrasses. Touchette, Brant W.; Burkholder, JoAnn M. Department of Botany, North Carolina State University, Raleigh, NC, USA. Journal of Experimental Marine Biology and Ecology (2000), 250(1-2), 133-167.
- Inorganic nitrogen utilization by assemblages of marine bacteria in seawater culture. Horrigan, S. G.; Hagstroem, A.; Koike, I.; Azam, F. Mar. Sci. Res. Cent., SUNY, Stony Brook, NY, USA. Marine Ecology: Progress Series (1988), 50(1-2), 147-50.
- Some observations on marine phytoplankton kinetics. 2. The effect of nitrate and ammonium concentrations on the growth and uptake rates of the natural population of Ubatuba region, SP (23°S, 045°W). Schmidt, Gilda. Inst. Oceanogr., Univ. Sao Paulo, Brazil. Boletim do Instituto Oceanografico (Universidade de Sao Paulo) (1983), 32(1), 83-90.
- Nutrient control of algal growth in estuarine waters. Nutrient limitation and the importance of nitrogen requirements and nitrogen storage among phytoplankton and species of macroalgae. Pedersen, Morten Foldager; Borum, Jens. Freshwater Biological Laboratory, University Copenhagen, Hillerod, Den. Marine Ecology: Progress Series (1996), 142(1 to 3), 261-272.
- Nutrient-enhanced growth of Cladophora prolifera in Harrington Sound, Bermuda: eutrophication of a confined, phosphorus-limited marine ecosystem. Lapointe, Brian E.; O’Connell, Julie. Harbor Branch Oceanogr. Inst., Inc., Big Pine Key, FL, USA. Estuarine, Coastal and Shelf Science (1989), 28(4), 347-60.
- Seasonal variations in eelgrass (Zostera marina L.) responses to nutrient enrichment and reduced light availability in experimental ecosystems. Moore, Kenneth A.; Wetzel, Richard L. The Virginia Institute of Marine Science, School of Marine Science, College of William and Mary, Gloucester Point, VA, USA. Journal of Experimental Marine Biology and Ecology (2000), 244(1), 1-28.
- Effects of nitrate and phosphate on growth and C2 toxin productivity of Alexandrium tamarense CI01 in culture. Wang, Da-Zhi; Hsieh, Dennis P. H. Department of Biology, The Hong Kong University of Science and Technology, Kowloon, Hong Kong SAR, Peop. Rep. China. Marine Pollution Bulletin (2002), 45(1-12), 286-289.
- Uptake and assimilation of dissolved inorganic nitrogen by a symbiotic sea anemone. Wilkerson, Frances P.; Muscatine, L. Dep. Biol., Univ. California, Los Angeles, CA, USA. Proceedings of the Royal Society of London, Series B: Biological Sciences (1984), 221(1222), 71-86.
- Nitrate assimilation by zooxanthellae maintained in laboratory culture. Wilkerson, F. P.; Trench, R. K. Dep. Biol. Sci., Univ. California, Santa Barbara, CA, USA. Marine Chemistry (1985), 16(4), 385-93.
- Captive Seawater Fishes : Science and Technology. Spotte, Stephen. (1992), 976 pp. Publisher: Interscience.
- Water quality requirements for first-feeding in marine fish larvae. I. Ammonia, nitrite, and nitrate. Brownell, Charles L. Dep. Zool., Univ. Cape Town, Rondebosch, S. Afr. Journal of Experimental Marine Biology and Ecology (1980), 44(2-3), 269-83.
- Nutrification impacts on coral reefs from northern Bahia, Brazil. Costa, O. S., Jr.; Leao, Z. M. A. N.; Nimmo, M.; Attrill, M. J. Plymouth Environmental Research Centre, University of Plymouth, Plymouth, UK. Hydrobiologia (2000), 440 307-315.
- Effects of lowered pH and elevated nitrate on coral calcification. Marubini, F.; Atkinson, M. J. Biosphere 2 Center, Columbia Univ., Oracle, AZ, USA. Marine Ecology: Progress Series (1999), 188 117-121.
- Bicarbonate addition promotes coral growth. Marubini, Francesca; Thake, Brenda. School of Biological Sciences, Queen Mary and Westfield College, London, UK. Limnology and Oceanography (1999), 44(3), 716-720.
- Nitrate increases zooxanthellae population density and reduces skeletogenesis in corals. Marubini, F.; Davies, P. S. Bellairs Research Inst., McGill University, St. James, Barbados. Marine Biology (Berlin) (1996), 127(2), 319-328.
- Nutrition of algal-invertebrate symbiosis. II. Effects of exogenous nitrogen sources on growth, photosynthesis and the rate of excretion by algal symbionts in vivo and in vitro. Taylor, D. L. Rosenstiel Sch. Mar. Atmos. Sci., Miami, FL, USA. Proceedings of the Royal Society of London, Series B: Biological Sciences (1978), 201(1145), 401-12.
- Tanaka, Yasuaki, Toshihiro Miyajima, Isao Koike, Takeshi Hayashibara, and Hiroshi Ogawa. “Imbalanced coral growth between organic tissue and carbonate skeleton caused by nutrient enrichment.” Limnology and oceanography 52, no. 3 (2007): 1139-1146.
- Effects of elevated seawater temperature and nitrate enrichment on the branching coral Porites cylindrica in the absence of particulate food Nordemar,I.; M Nyström, M.; Dizon, R. Marine Biology (2003) 142:669-677.
- An introduction to the chemistry of the sea. Pilson, Michael E. Q. (1998) 431 pp. Publisher: Pearson Education POD.
- Nitrate Removal — A New Alternative by Randy Donowitz, Aquarium Frontiers April 1998. http://www.animalnetwork.com/fish2/aqfm/1998/april/features/2/default.asp

Leave a Reply
You must be logged in to post a comment.